Seznamy 85 Atom Diagram With Protons Neutrons And Electrons Výborně
Seznamy 85 Atom Diagram With Protons Neutrons And Electrons Výborně. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.
Tady Sub Atomic Particles Chemistry Libretexts
The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. The atomic number of an element describes the total number of protons in its nucleus.In chemical reactions, atoms are combined, separated or rearranged.
If there are more protons than electrons, an atomic ion has a positive charge and is called a … Electrons of all the elements. Neutral atoms have equal numbers of protons and electrons. • protons have a positive charge. This one shows the protons, neutrons, and electrons of a carbon atom.

• protons and neutrons are in the center of the atom, making up the nucleus... Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.

However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. . • protons and neutrons are in the center of the atom, making up the nucleus.

If there are more protons than electrons, an atomic ion has a positive charge and is called a ….. If there are more protons than electrons, an atomic ion has a positive charge and is called a … In chemical reactions, atoms are combined, separated or rearranged. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. Neutral atoms have equal numbers of protons and electrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. This one shows the protons, neutrons, and electrons of a carbon atom.
Electrons of all the elements. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. An ion of an atom is one in which the number of protons and electrons is not the same. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. • electrons have a negative charge. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. This one shows the protons, neutrons, and electrons of a carbon atom. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. • protons have a positive charge. • protons and neutrons are in the center of the atom, making up the nucleus. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.
The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.. Neutral atoms have equal numbers of protons and electrons. /captionthe image on the left is a basic atom diagram. • electrons have a negative charge. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. If there are more protons than electrons, an atomic ion has a positive charge and is called a … In chemical reactions, atoms are combined, separated or rearranged. • electrons surround the nucleus. • protons and neutrons are in the center of the atom, making up the nucleus. The atomic number of an element describes the total number of protons in its nucleus. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons.. Neutral atoms have equal numbers of protons and electrons.

Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.
• electrons surround the nucleus. This one shows the protons, neutrons, and electrons of a carbon atom. In chemical reactions, atoms are combined, separated or rearranged. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. • protons and neutrons are in the center of the atom, making up the nucleus. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. /captionthe image on the left is a basic atom diagram.. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.

• electrons have a negative charge.. • electrons surround the nucleus. If there are more protons than electrons, an atomic ion has a positive charge and is called a … However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. This one shows the protons, neutrons, and electrons of a carbon atom. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.. • protons have a positive charge.

112 sor · 01/11/2021 · protons, neutrons and.. /captionthe image on the left is a basic atom diagram. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. Neutral atoms have equal numbers of protons and electrons. • electrons have a negative charge.

If there are more protons than electrons, an atomic ion has a positive charge and is called a … This one shows the protons, neutrons, and electrons of a carbon atom. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. • protons and neutrons are in the center of the atom, making up the nucleus. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. • electrons have a negative charge. • protons have a positive charge. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion.

In chemical reactions, atoms are combined, separated or rearranged. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. An ion of an atom is one in which the number of protons and electrons is not the same. 112 sor · 01/11/2021 · protons, neutrons and. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. Neutral atoms have equal numbers of protons and electrons. /captionthe image on the left is a basic atom diagram. In chemical reactions, atoms are combined, separated or rearranged. • electrons have a negative charge. In chemical reactions, atoms are combined, separated or rearranged.

If there are more protons than electrons, an atomic ion has a positive charge and is called a … The atomic number of an element describes the total number of protons in its nucleus. Electrons of all the elements. This one shows the protons, neutrons, and electrons of a carbon atom. If there are more protons than electrons, an atomic ion has a positive charge and is called a … • electrons have a negative charge. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. /captionthe image on the left is a basic atom diagram. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion.

3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom... In chemical reactions, atoms are combined, separated or rearranged. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. /captionthe image on the left is a basic atom diagram. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. • electrons have a negative charge. In chemical reactions, atoms are combined, separated or rearranged.

The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom.. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.

Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • protons have a positive charge. • electrons surround the nucleus. An ion of an atom is one in which the number of protons and electrons is not the same. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons... Neutral atoms have equal numbers of protons and electrons.

• protons have a positive charge. In chemical reactions, atoms are combined, separated or rearranged. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. • protons have a positive charge. The atomic number of an element describes the total number of protons in its nucleus. Electrons of all the elements. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. An ion of an atom is one in which the number of protons and electrons is not the same. 112 sor · 01/11/2021 · protons, neutrons and.

/captionthe image on the left is a basic atom diagram. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. • electrons surround the nucleus. If there are more protons than electrons, an atomic ion has a positive charge and is called a … Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. • protons have a positive charge. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. Electrons of all the elements. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. 112 sor · 01/11/2021 · protons, neutrons and. An ion of an atom is one in which the number of protons and electrons is not the same. /captionthe image on the left is a basic atom diagram.

• protons and neutrons are in the center of the atom, making up the nucleus. /captionthe image on the left is a basic atom diagram. Electrons of all the elements. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. • electrons have a negative charge. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. The atomic number of an element describes the total number of protons in its nucleus.. • electrons have a negative charge.

• protons and neutrons are in the center of the atom, making up the nucleus. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. This one shows the protons, neutrons, and electrons of a carbon atom. • electrons have a negative charge.

Electrons of all the elements.. If there are more protons than electrons, an atomic ion has a positive charge and is called a …. • electrons surround the nucleus.
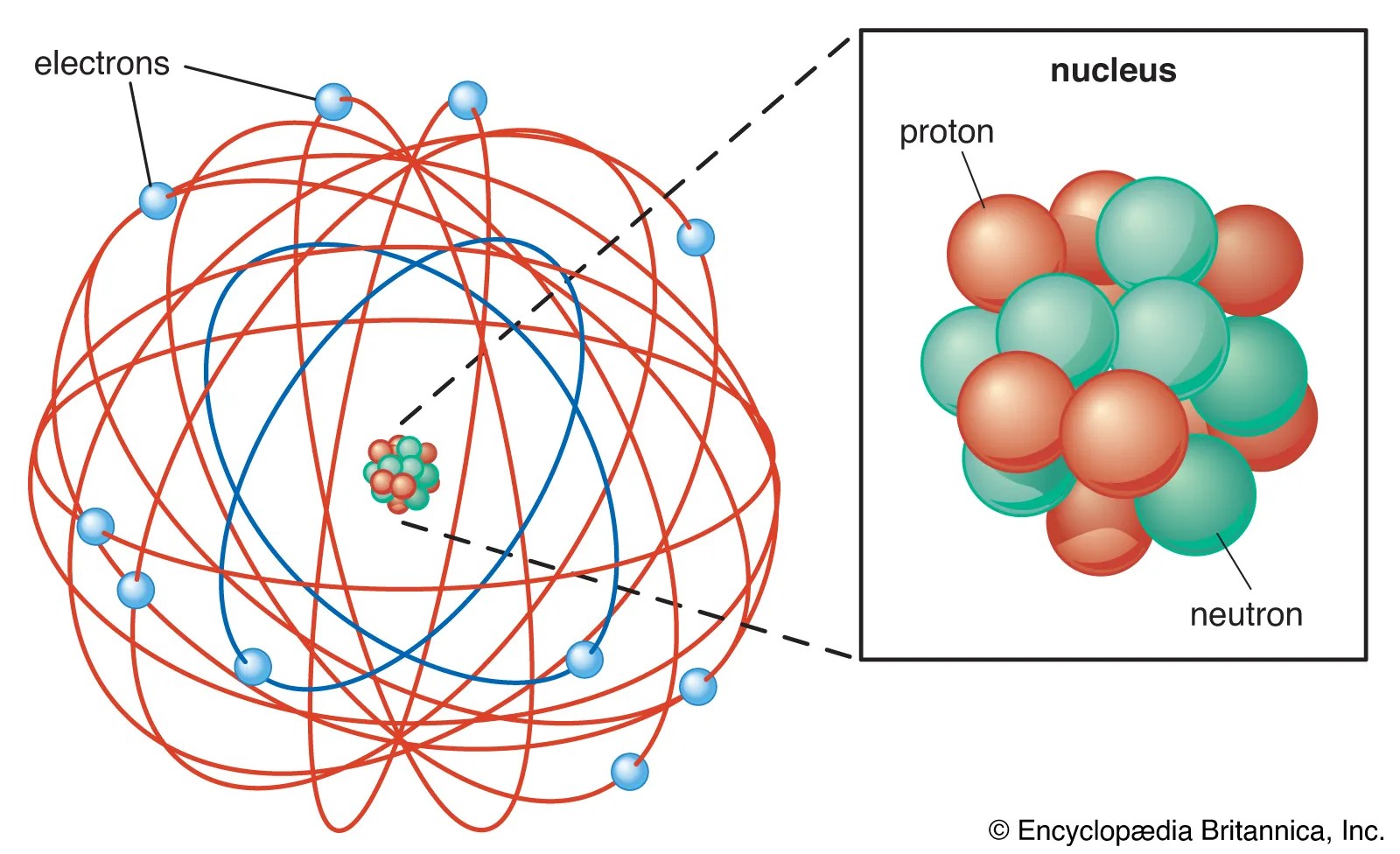
Electrons of all the elements.. 112 sor · 01/11/2021 · protons, neutrons and. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.

An ion of an atom is one in which the number of protons and electrons is not the same.. In chemical reactions, atoms are combined, separated or rearranged... Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.

3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.. An ion of an atom is one in which the number of protons and electrons is not the same. • protons have a positive charge... Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. .. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.

Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.. 112 sor · 01/11/2021 · protons, neutrons and.

The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. Neutral atoms have equal numbers of protons and electrons. • electrons surround the nucleus. • protons have a positive charge. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. An ion of an atom is one in which the number of protons and electrons is not the same. • protons and neutrons are in the center of the atom, making up the nucleus. Electrons of all the elements. 112 sor · 01/11/2021 · protons, neutrons and. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons... Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.

Electrons of all the elements. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. This one shows the protons, neutrons, and electrons of a carbon atom. /captionthe image on the left is a basic atom diagram. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. The atomic number of an element describes the total number of protons in its nucleus. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. An ion of an atom is one in which the number of protons and electrons is not the same. 112 sor · 01/11/2021 · protons, neutrons and. If there are more protons than electrons, an atomic ion has a positive charge and is called a …

/captionthe image on the left is a basic atom diagram. An ion of an atom is one in which the number of protons and electrons is not the same. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • electrons have a negative charge.

• electrons surround the nucleus... An ion of an atom is one in which the number of protons and electrons is not the same. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. • electrons surround the nucleus. The atomic number of an element describes the total number of protons in its nucleus. • protons have a positive charge. /captionthe image on the left is a basic atom diagram. Neutral atoms have equal numbers of protons and electrons. Electrons of all the elements. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not... The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.

112 sor · 01/11/2021 · protons, neutrons and. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. • protons have a positive charge. Electrons of all the elements. /captionthe image on the left is a basic atom diagram. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. The atomic number of an element describes the total number of protons in its nucleus... • protons have a positive charge.

112 sor · 01/11/2021 · protons, neutrons and. Neutral atoms have equal numbers of protons and electrons. • protons have a positive charge.

Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. The atomic number of an element describes the total number of protons in its nucleus. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. • electrons surround the nucleus. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. • protons and neutrons are in the center of the atom, making up the nucleus. If there are more protons than electrons, an atomic ion has a positive charge and is called a … The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.

Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons... An ion of an atom is one in which the number of protons and electrons is not the same. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The atomic number of an element describes the total number of protons in its nucleus. • electrons have a negative charge. /captionthe image on the left is a basic atom diagram. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. Neutral atoms have equal numbers of protons and electrons. 112 sor · 01/11/2021 · protons, neutrons and. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. In chemical reactions, atoms are combined, separated or rearranged. • electrons surround the nucleus.

If there are more protons than electrons, an atomic ion has a positive charge and is called a …. In chemical reactions, atoms are combined, separated or rearranged. The atomic number of an element describes the total number of protons in its nucleus. An ion of an atom is one in which the number of protons and electrons is not the same. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • protons have a positive charge. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion... • protons have a positive charge.

Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.. • protons and neutrons are in the center of the atom, making up the nucleus. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. /captionthe image on the left is a basic atom diagram. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons.

Neutral atoms have equal numbers of protons and electrons. • protons and neutrons are in the center of the atom, making up the nucleus. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom.

If there are more protons than electrons, an atomic ion has a positive charge and is called a ….. • electrons surround the nucleus. Neutral atoms have equal numbers of protons and electrons.. Neutral atoms have equal numbers of protons and electrons.

Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge... The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. This one shows the protons, neutrons, and electrons of a carbon atom.. • electrons surround the nucleus.

• protons have a positive charge. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • electrons surround the nucleus. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. • protons have a positive charge. Neutral atoms have equal numbers of protons and electrons. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom.. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom.

• protons and neutrons are in the center of the atom, making up the nucleus... In chemical reactions, atoms are combined, separated or rearranged. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge... Electrons of all the elements.

3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. /captionthe image on the left is a basic atom diagram. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. This one shows the protons, neutrons, and electrons of a carbon atom. • electrons surround the nucleus. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.

112 sor · 01/11/2021 · protons, neutrons and. In chemical reactions, atoms are combined, separated or rearranged. • electrons surround the nucleus. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. The atomic number of an element describes the total number of protons in its nucleus. This one shows the protons, neutrons, and electrons of a carbon atom. /captionthe image on the left is a basic atom diagram. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge.

20/07/2016 · remember, a neutral atom contains the same number of protons and electrons.. The atomic number of an element describes the total number of protons in its nucleus. • electrons surround the nucleus. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. Electrons of all the elements.

The atomic number of an element describes the total number of protons in its nucleus. An ion of an atom is one in which the number of protons and electrons is not the same. /captionthe image on the left is a basic atom diagram. 112 sor · 01/11/2021 · protons, neutrons and.

Neutral atoms have equal numbers of protons and electrons. • electrons surround the nucleus. In chemical reactions, atoms are combined, separated or rearranged. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. If there are more protons than electrons, an atomic ion has a positive charge and is called a … • protons have a positive charge. This one shows the protons, neutrons, and electrons of a carbon atom.. If there are more protons than electrons, an atomic ion has a positive charge and is called a …

The atomic number of an element describes the total number of protons in its nucleus. In chemical reactions, atoms are combined, separated or rearranged. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. An ion of an atom is one in which the number of protons and electrons is not the same.

• electrons surround the nucleus.. In chemical reactions, atoms are combined, separated or rearranged. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. This one shows the protons, neutrons, and electrons of a carbon atom. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. The atomic number of an element describes the total number of protons in its nucleus. 112 sor · 01/11/2021 · protons, neutrons and. • electrons have a negative charge. • electrons surround the nucleus. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. Electrons of all the elements. In chemical reactions, atoms are combined, separated or rearranged.

• electrons have a negative charge. • electrons have a negative charge. Electrons of all the elements. • protons and neutrons are in the center of the atom, making up the nucleus. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. /captionthe image on the left is a basic atom diagram. Neutral atoms have equal numbers of protons and electrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. This one shows the protons, neutrons, and electrons of a carbon atom. An ion of an atom is one in which the number of protons and electrons is not the same. The atomic number of an element describes the total number of protons in its nucleus.

Neutral atoms have equal numbers of protons and electrons. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. • electrons have a negative charge. This one shows the protons, neutrons, and electrons of a carbon atom. • electrons surround the nucleus. 112 sor · 01/11/2021 · protons, neutrons and. The atomic number of an element describes the total number of protons in its nucleus. Electrons of all the elements. • protons have a positive charge.. If there are more protons than electrons, an atomic ion has a positive charge and is called a …

In chemical reactions, atoms are combined, separated or rearranged.. This one shows the protons, neutrons, and electrons of a carbon atom. /captionthe image on the left is a basic atom diagram. • electrons surround the nucleus. • protons have a positive charge.

112 sor · 01/11/2021 · protons, neutrons and... Electrons of all the elements. • protons have a positive charge. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. An ion of an atom is one in which the number of protons and electrons is not the same. • electrons surround the nucleus. Neutral atoms have equal numbers of protons and electrons. • electrons have a negative charge. • protons and neutrons are in the center of the atom, making up the nucleus.

Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. • electrons have a negative charge. 112 sor · 01/11/2021 · protons, neutrons and.

• electrons have a negative charge... The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. • protons and neutrons are in the center of the atom, making up the nucleus. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. The atomic number of an element describes the total number of protons in its nucleus. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • protons have a positive charge. If there are more protons than electrons, an atomic ion has a positive charge and is called a … • electrons have a negative charge.. • electrons surround the nucleus.

The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. In chemical reactions, atoms are combined, separated or rearranged. Neutral atoms have equal numbers of protons and electrons. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.

Electrons of all the elements.. An ion of an atom is one in which the number of protons and electrons is not the same. This one shows the protons, neutrons, and electrons of a carbon atom. In chemical reactions, atoms are combined, separated or rearranged. If there are more protons than electrons, an atomic ion has a positive charge and is called a … • electrons have a negative charge. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • protons and neutrons are in the center of the atom, making up the nucleus. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.. If there are more protons than electrons, an atomic ion has a positive charge and is called a …

Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. If there are more protons than electrons, an atomic ion has a positive charge and is called a … 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. The atomic number of an element describes the total number of protons in its nucleus. • electrons have a negative charge.. Electrons of all the elements.

3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. /captionthe image on the left is a basic atom diagram. In chemical reactions, atoms are combined, separated or rearranged. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. The atomic number of an element describes the total number of protons in its nucleus. • electrons surround the nucleus. An ion of an atom is one in which the number of protons and electrons is not the same. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it.. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.

• electrons have a negative charge.. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. 112 sor · 01/11/2021 · protons, neutrons and. Neutral atoms have equal numbers of protons and electrons. • protons and neutrons are in the center of the atom, making up the nucleus. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.. Electrons of all the elements.

• electrons have a negative charge... The atomic number of an element describes the total number of protons in its nucleus. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. • protons and neutrons are in the center of the atom, making up the nucleus. This one shows the protons, neutrons, and electrons of a carbon atom. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. The atomic model in the diagram below shows protons and neutrons concentrated at the atomic nucleus and electrons in the orbits surrounding it. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion.

• electrons have a negative charge... This one shows the protons, neutrons, and electrons of a carbon atom. Electrons of all the elements. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. • electrons surround the nucleus. However, atoms may gain or lose electrons in order to increase their stability and the resulting charged entity is called an ion. In chemical reactions, atoms are combined, separated or rearranged. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. 20/07/2016 · remember, a neutral atom contains the same number of protons and electrons.. An ion of an atom is one in which the number of protons and electrons is not the same.

3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom. Protons are positively charged, electrons are negatively charged, while the neutrons carry no charge. This one shows the protons, neutrons, and electrons of a carbon atom. Electrons of all the elements. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. 3 sor · you can use these numbers to calculate the number of protons, neutrons and electrons in an atom.

The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not.. /captionthe image on the left is a basic atom diagram.. • protons have a positive charge.